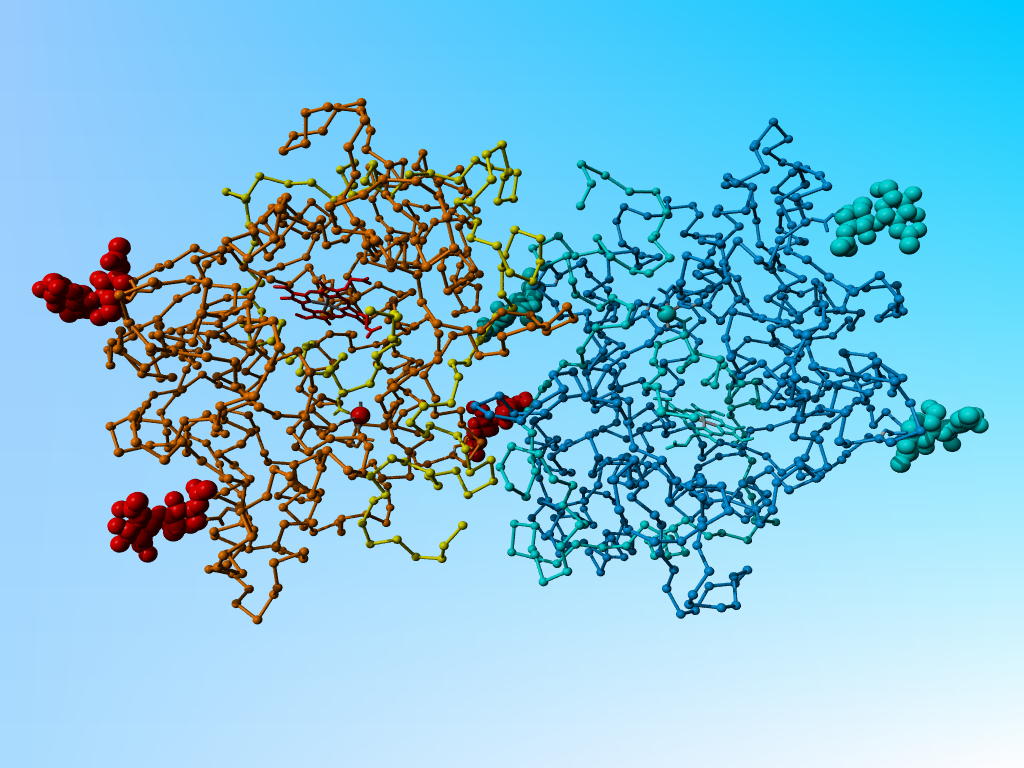
1myp trace diagram. Chains A and C (and their ligands in yellow, brown and red respectively. Chains B, D, and their ligands in blueish. Click on the picture for a short movie.
JRNL AUTH J.ZENG,R.E.FENNA 1MYP 7 JRNL TITL X-RAY CRYSTAL STRUCTURE OF CANINE MYELOPEROXIDASE 1MYP 8 JRNL TITL 2 AT 3 ANGSTROMS RESOLUTION 1MYP 9 JRNL REF J.MOL.BIOL. V. 226 185 1992 1MYP 10 JRNL REFN ASTM JMOBAK UK ISSN 0022-2836 070 1MYP 11 |
This molecule is a dimer. A BLAST alignment of the chains B and D versus the SwissProt database is shown as supplemental material:
Supplemental materialTogether with their annotation:
REMARK 4 THE MOLECULE IS A DIMER WITH EXACT OR NEAR-EXACT TWO-FOLD 1MYP 23 REMARK 4 SYMMETRY. THE 2 HALVES ARE COVALENTLY LINKED BY A SINGLE 1MYP 24 REMARK 4 DISULFIDE AT CYS 153. ONE HALF OF THE MOLECULE IS 1MYP 25 REMARK 4 COMPRISED OF RESIDUES A 1- A 104, C 114 - C 575, 1MYP 26 REMARK 4 HEM A 580, CA A 581, AND SIX NAG'S, A 582 - A 587. 1MYP 27 REMARK 4 THE OTHER HALF CONSISTS OF RESIDUES B 1 - B 104, D 114 - 1MYP 28 REMARK 4 D 575, HEM B 580, CA B 581, AND SIX NAG'S, B 582 - B 587. 1MYP 29 |
it becomes clear that the chain-ids are weirdly chosen. The order of the chain-ids in the molecule is A, C, B, D.

|
1myp trace diagram. Chains A and C (and their ligands in yellow, brown and red respectively. Chains B, D, and their ligands in blueish. Click on the picture for a short movie. |
However, this is not yet the most worrysom aspect of 1myp. Look at the chemical bonds between Asn 225 A and its Nag. That includes a funny oxygen-oxygen bond.
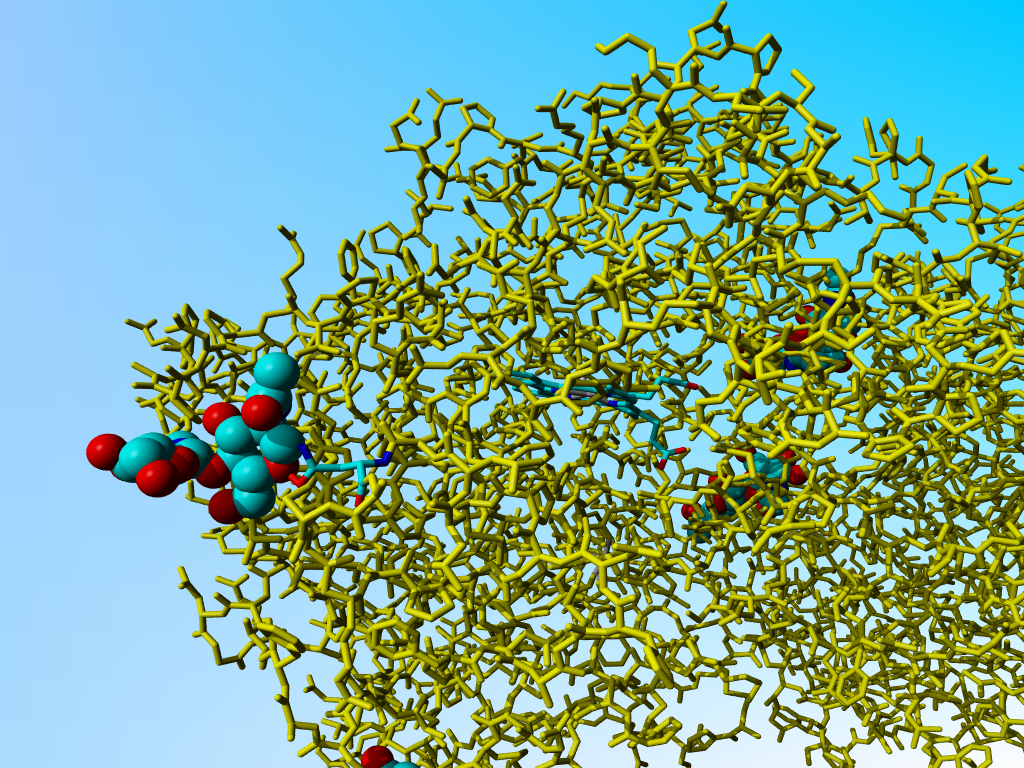
|
The Nag - Asn 225 A bonds. All protein in yellow, except Asn 225. Click on the figure for a zoom-in. |
This is even funnier as they state above that the two halves share near-exact two-fold symmetry, and the the symmetry partners in chain B and D are fine:
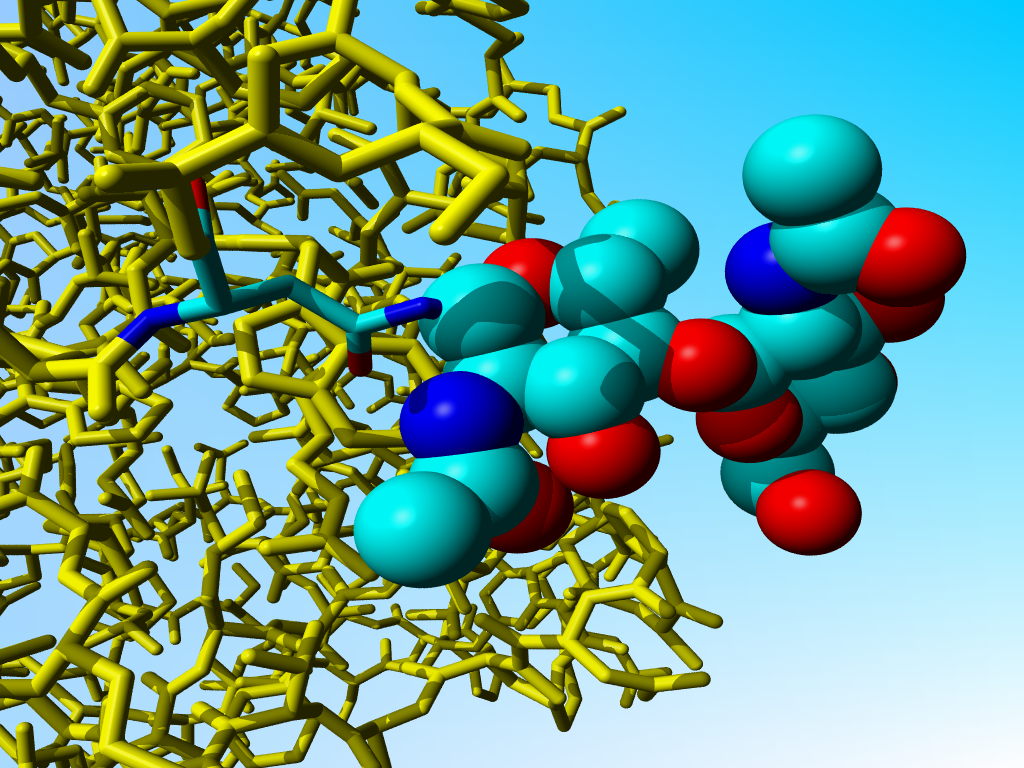
|
Symmetry partner of the Nag - Asn shown above. |